Health officials have urged football fans to celebrate the Super Bowl at home this year. The virus is indifferent to whether people cheer for the Tampa Bay Buccaneers or the Kansas City Chiefs, or just enjoy the commercials and the halftime show. Some will heed this advice, while others may be unable to resist watching the biggest football game of the year with their fellow fans.
Of course, the decision between watching at home and watching with a group of friends and family comes with the risk of COVID-19 exposure. Experts increasing point to small gatherings in homes and apartments as one of the leading contributors to the ongoing spread of COVID-19.
For those who watch the Big Game with a group of friends and are exposed to COVID-19, isolation isn’t enough. It is time to be part of the solution.
‘I think I was exposed‘
Even though highly effective vaccines are being administered, people will continue to test positive for the foreseeable future. New COVID-19 variants are also a cause for concern. If you think you might have been exposed to COVID-19, rather than take a “wait and see” approach or remain silent out of potential embarrassment, consider joining a clinical trial.
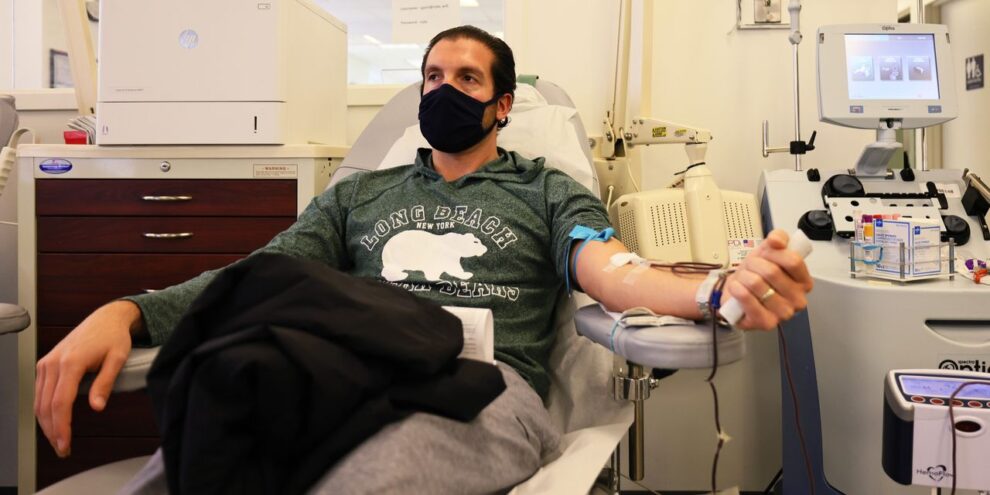
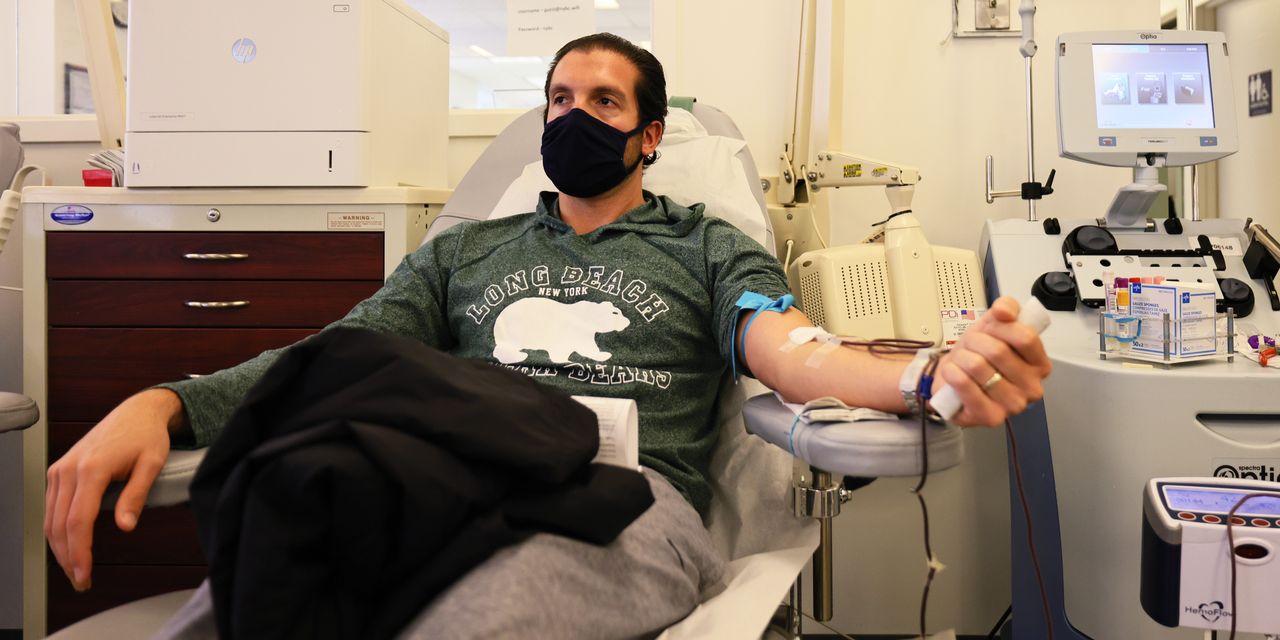
My colleagues and I at John Hopkins University are working with 25 testing sites in major U.S. cities and in the Navajo Nation on a potentially game-changing solution that could help us reduce COVID-19 hospitalizations and control its spread. We are investigating whether a blood plasma transfusion containing high levels of antibodies to COVID-19, given early in illness, reduces the severity of the disease or even prevents people from developing an infection entirely.
For those at high-risk, we believe this treatment has the potential to cut hospitalizations by half and prevent deaths. For those with milder cases, we believe that antibody-rich plasma will speed up recovery time and reduce the spread of the virus. But to really know, we must do the clinical trials.
While a vaccine may be quicker than getting a blood transfusion, convalescent plasma is perhaps our best short-term hope. Consider the impact on a Big Game party where one attendee tested positive for COVID-19. Instead of each person who went to the gathering having to quarantine for two weeks, both the infected individual and those who came into contact with that person could immediately be given a dose of plasma, possibly creating a circle of immunity around a positive case and preventing the virus from spreading.
Now apply that to a high school, where a student tested positive. A poultry processing line at a meat factory. An aircraft carrier. A nursing home.
How convalescent plasma works
Convalescent plasma is not a new therapy. Physicians have used blood plasma for more than a century to combat severe diseases in hospitals, including, for example, measles — a disease even more transmissible than COVID-19 — as well as childhood diphtheria infection, the influenza pandemic of 1918 and the 2003 outbreak of severe acute respiratory syndrome. This treatment was even awarded the Noble Prize in 1901 for the lifesaving nature of antibody transfer in childhood diphtheria infection. Now we are applying modern blood banking technology, the latest in immunology and virology science and clinical trial methodology, to find out if, when and how it works against COVID-19.
When compared to drugs or vaccines, the potential benefits of blood plasma are many — it is made in nature, not in factories, therefore the chances of supply chain issues or factory recalls are lower. Production, delivery and storage are low-cost and cost-effective, and it can be easily scaled, especially in resource-constrained countries such as India and Brazil.
If approved, outpatient clinics with proper freezer storage and sterilization for blood transfusions could immediately treat people anywhere in the world. Additionally, plasma from individuals who have defeated the viral variants can be quickly implemented to respond to this threat more quickly than monoclonals or vaccines.
While I can’t yet claim this treatment will work, there is reason for hope. A recent Argentinian study published in The New England Journal of Medicine of 160 patients, all over the age of 65, showed that the early use of convalescent plasma reduced the relative risk of severe respiratory disease by 48% in the adults being studied.
If Johns Hopkins’ trials can confirm the use of blood plasma is effective in all age groups, we would be that much closer to preventing outbreaks, reducing periods of transmission, lowering hospitalizations and deaths, and getting back to normal, sooner, so that we can all root for our favorite teams alongside our friends and family, without risking our health and safety.
How to help
Here’s where you come in. If you have tested positive for COVID-19 in the past six (6) days or have been exposed in the last three days (3) to a family member or colleague who has tested positive, you could potentially receive a life-saving treatment and help end the pandemic. You can use this Clinical Trial Location Finder to find the testing site closest to you.
On the first day of joining our study, a volunteer will be enrolled in the trial and receive a physical exam. The next day, the volunteer will receive a transfusion of plasma. As the illness progresses, the volunteer will attend virtual and in-person follow-up visits. Volunteers will have direct access to COVID-19 testing and top-quality infectious disease doctors as they go through the phases of recovery. All medical care provided as part of the study is free, and volunteers may be compensated.
Watching the Super Bowl has become an unofficial holiday in the U.S. It’s a day when we can come together with friends and family to share snacks over sports, watch extravagant commercials and see celebrity musical acts at halftime. While COVID-19 might have the power to take these traditions away from us right now, that can all change if we come together and fight for ways to regain what we value.
Shmuel Shoham, M.D., is an infectious disease expert and Associate Professor of Medicine at the Johns Hopkins School of Medicine and a principal investigator for the Johns Hopkins Plasma Trials. To see if you qualify for the study, visit CovidPlasmaTrial.org or call 888-506-1199.
Also read: Dr. Fauci warns against Super Bowl parties: ‘Just lay low and cool it’